Background:
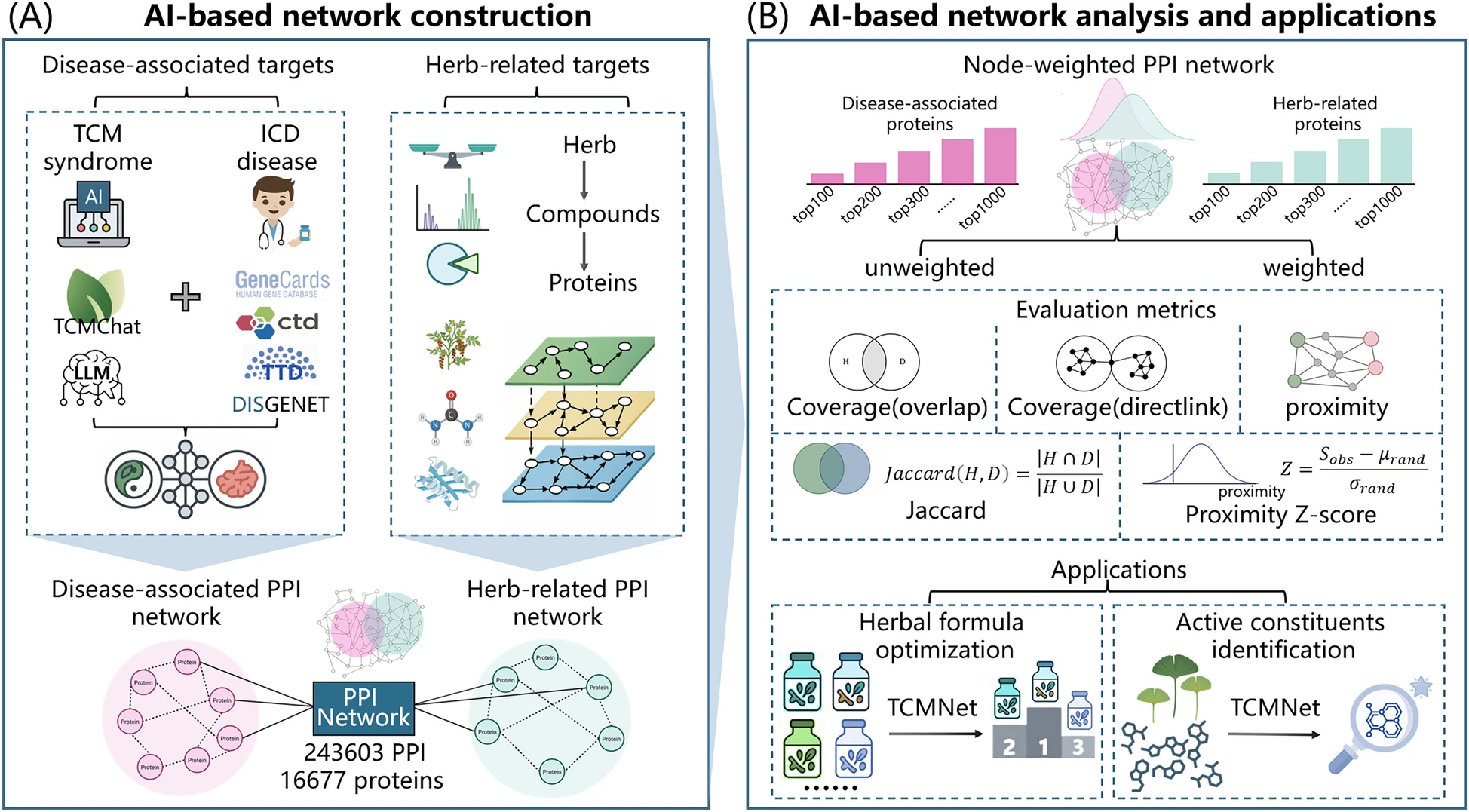
Artificial intelligence (AI), particularly large language models (LLMs), have provided powerful tools for systematically modeling the complexity of traditional Chinese medicine (TCM). To overcome the limitations of subjective formula design and unweighted target prioritization, we developed TCMNet, an AI-powered strategy that integrates LLM-assisted disease knowledge mining, protein–protein interaction (PPI) networks, and deep learning-based binding prediction to support herbal formula evaluation and active compounds identification.
Methods:
TCMNet integrates AI-guided literature analysis with weighted PPI network evaluation. Parkinson’s disease (PD) was chosen as the representative case study. Disease-associated protein targets were semantically weighted using TCMChat, a TCM-specific LLM that extracts relevant targets from Chinese and English literature. Furthermore, herb-specific information data, such as composition ratios, compound abundance, and compound-protein interaction probabilities, was used to generate weighted herb-related proteins. These weights were incorporated into a PPI network to assign biological weight to each node. Four classical TCM formulas (Tianma Gouteng Decoction, Liuwei Dihuang, Qianzheng San, Dabuyin Wan), the clinically optimized Pingchan Granule (PCG), and their integrative combinations with Western medicine (Levodopa) were systematically evaluated alongside the single-herb Ginkgo biloba. Therapeutic relevance was assessed using network-based metrics such as target coverage, Jaccard similarity, and weighted proximity, with statistical significance measured by Z-scores. To validate key active compounds, we employed Boltz-2, a state-of-the-art deep learning method, to predict the binding probabilities between herbal compounds and prioritized PD-associated proteins.
Results:
Weighted proximity metrics markedly outperformed unweighted measures across all four evaluated TCM formulas, demonstrating the substantial benefit of integrating node weights. Among the evaluated formulas, Tianma Gouteng Decoction demonstrated superior performance in target coverage and network proximity, aligning well with existing literature. Computational validation on Pingchan Granule (PCG) confirmed that TCMNet successfully captures the therapeutic retention of formula optimization. Furthermore, integrative strategies combining TCM with Levodopa exhibited significantly enhanced network proximity compared to monotherapy, supporting the rationale for combined treatment. Moreover, the case study identified flavonoids and isoflavonoids from Ginkgo biloba as the primary bioactive constituents contributing to anti-PD activity. Boltz-2 deep learning predictions further confirmed that flavonoid compounds exhibited significantly higher binding probabilities and affinities toward key PD-associated proteins compared to non-flavonoids, thus validating the results of TCMNet.
Conclusions:
By explicitly incorporating protein weights through combining LLM-guided target identification with node-weighted evaluation, TCMNet offers a new AI-driven strategy for optimizing TCM. This approach enables herbal formula evaluation and optimization as well as the identification of bioactive constituents, advancing the modernization of herbal medicine research.